CH 3 2 CO. What is the boiling point of RBF.

Is Hbr Polar Or Nonpolar Hydrogen Bromide Youtube
They will increase in strength depending on the number of electrons around the molecules.

. Therefore it has dipole-dipole forces. CCl4 is nonpolar in nature but its bond is polar. The bond angle of CCl4 is 1095º.
Each HBr molecule is attracted to other HBr molecules by a mixture of permanent dipole-dipole and dispersion. HBr is a polar molecule. There are also dispersion forces between HBr molecules.
This is a very specific bond as indicated. In this compound the carbon atom bonded to the oxygen atom has a charge of partial positive δ. What is the molecular geometry of CCl4.
HBr is a polar molecule. Does HBr have London dispersion forces as its only intermolecular force. Here all the hydrogen halides will have instantaneous dipole_induced dipole forces.
CH3OH and CH3CH2OH have hydrogen bonds due to the very electronegative O atom bonded to the H atom. H-Br is a polar covalent molecule with intramolecular covalent bonding. 2566F 1408C Why does CH3OH have a higher boiling point.
Does n2 have hydrogen bonding. And oxygen has a charge of partial negative δ-. CCl4 molecular geometry is tetrahedral and its electron geometry is also tetrahedral.
This compound has the next highest boiling point. There are also dispersion forces between HBr molecules. But permanent dipole-dipole forces are found between polar molecules because their dipole is permanent.
Nitrogen has only one lone pair only one hydrogen bond can be made to each nitrogen. Lets take propanone as an example. The larger halides will have more electrons so you would expect HI then HBr then HCl to have the highest bps as I atoms are largest and Cl atoms are smallest.
No since its a polar compound its also considered to be polar. Dispersion forces present in all matter and dipole-dipole forces will be present.

Best Overview Is Hbr Polar Or Nonpolar Science Education And Tutorials
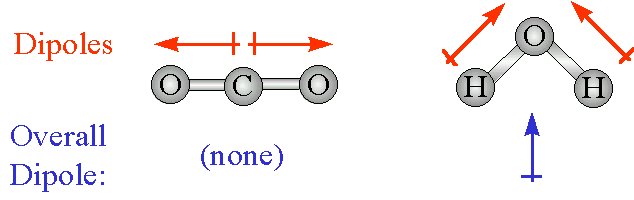
Which Of The Following Compounds Exhibits Only Dispersion And Dipole Dipole Intermolecular Interactions A Hbr B Co2 C H2o D N2 Socratic

0 Comments